|
3/20/2024 0 Comments Atom model bohr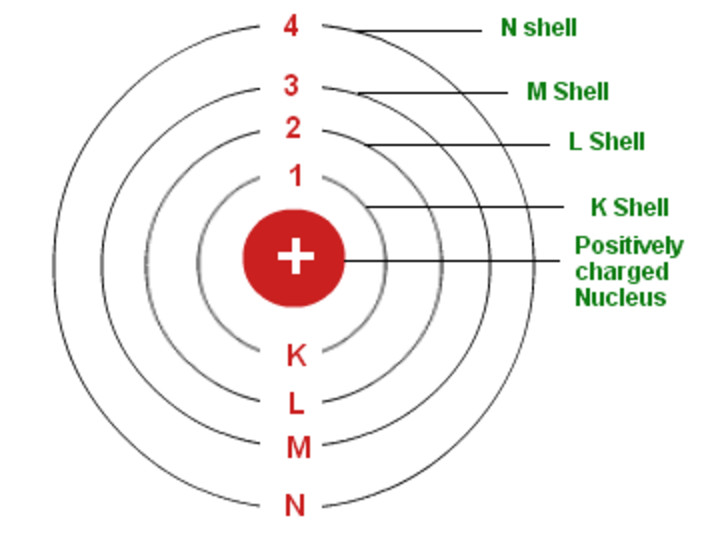
1 I denne model er atomet arrangeret som en atomkerne med en positiv ladning, omkredset af negative elektroner, på stort set samme måde som solsystemet, men med elektrostatiske tiltrækningskræfter i stedet for massetiltrækning. Check how the prediction of the model matches the experimental results. It does introduce several important features of all models used to describe the distribution of electrons in an atom. How did scientists figure out the structure of atoms without looking at them Try out different models by shooting light at the atom. Bohr’s model explained why atoms only emit light. Electrons move around a nucleus, but only in prescribed orbits, and If electrons jump to a lower-energy orbit, the difference is sent out as radiation. Instead of allowing for continuous values for the angular momentum, energy, and orbit radius, Bohr assumed that only discrete values for these could occur (actually, quantizing any one of these would imply that the other two are also quantized). An early model of the atom was developed in 1913 by Danish scientist Niels Bohr (18851962). Bohrs atommodel fra 1913 blev formuleret af den danske fysiker Niels Bohr. Bohr’s model of the hydrogen atom provides insight into the behavior of matter at the microscopic level, but it does not account for electronelectron interactions in atoms with more than one electron. In 1913, Niels Bohr proposed a theory for the hydrogen atom, based on quantum theory that some physical quantities only take discrete values.  
The absolute value of the energy difference is used, since frequencies and wavelengths are always positive. In this equation, h is Planck’s constant and E i and E f are the initial and final orbital energies, respectively.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
